Next generation CRISPR diagnostics
New advancements in CRISPR technology has led to the creation of next generation diagnostics, capable of detecting any nucleic acid motif and turning it into a signal that can be read without laboratory equipment. HOLMES uses Cas12a to produce a fluorescent signal and CRISPR–Chip uses Cas9 to trigger an electric signal directly.
However, the diagnostic accuracy of those approaches depends on how discriminative the chosen CRISPR target site is. Sites need to be highly specific and not bind to similar DNA sequences to prevent false positives. This is of particular relevance when trying to distinguish between similar viral strains, such as influenza strains. On the other hand, sites also need to be very active to avoid false negatives and have clinically relevant sensitivity.
Researchers must therefore balance selecting effective target sites with selecting unique target sites.
Using computational tools for design
Computational optimisation methods can be used to assist in the design of CRISPR targets for diagnosis. Researchers at CSIRO have developed TUSCAN, a high throughput tool for identifying and evaluating the potential effectiveness of CRISPR targets. Available as a cloud service as part of the GT-Scan suite, this tool is powerful enough to rank potential target sites across an entire genome. To identify unique target sites for diagnostic tools, researchers can compare target site candidates between different viruses or strains to optimise for effectiveness (sensitivity) as well as uniqueness (specificity).
Case study: diagnosing avian and swine influenza
As a proof of principle, we sought to design discriminative targets between two related influenza strains: H1N1 (swine flu) and H5N1 (bird flu), both of which were isolated from patients from Thailand in 2006. These two strains can have widely different clinical outcomes, meaning correct diagnosis is critical.
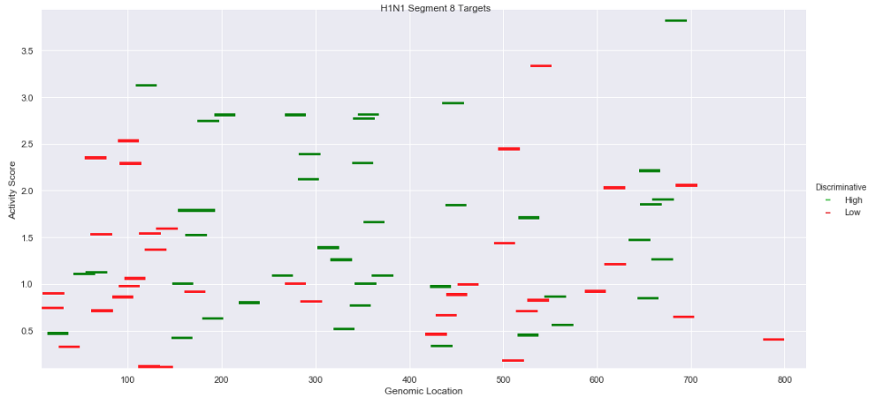
In this notebook, we used TUSCAN to identify and evaluate every potential CRISPR target site across both viral strains. Comparison between the list of target sites then let us identify which were unique to each virus.
This approach can be readily applied to any pathogen or combination of pathogens to identify unique targets for diagnosis by using just this repository.


